AtTechStar, we believe great measurement starts with the right technology and ends with solutions that are reliable, scalable, and supported long after startup. That’s why we’re excited to expand our analytical portfolio through our representation of Process Insights, bringing together a broad range of proven analytical technologies.
TechStar Expands Analytical Capabilities with Process Insights Solutions
Topics: Analytical Solutions, Analyzer, Process Insights
Wastewater treatment plants (also called Water Resource Facility WRFF) must treat the water before it can be released back into the environment (rivers, lakes, etc) and before it can be used as drinking water for a city, town, or municipality.
Topics: Analytical Solutions, Wastewater, Analyzer, KECO
How Waltron’s Instrumentation Products Helped A Semiconductor Manufacturer Detect A Leak
For years, a semiconductor manufacturer had been dealing with unreliable Dissolved Oxygen Analyzers from one of Waltron’s competitors. The maintenance and lay-up procedures were inconvenient and time-consuming.
Topics: Analytical Solutions, Analyzer, Water Chemistry, Waltron
Simplify Your Maintenance with Waltron’s Easy-to-Use Luminescent Dissolved Oxygen Analyzers
The process of monitoring water and steam quality in industrial facilities like power plants and paper mills is often very complex and requires much expertise. However, maintaining these instruments can be an even bigger challenge. If you are struggling with instruments that are too complicated to maintain, Waltron’s water and steam quality monitoring instruments might be just what you need to simplify your maintenance.
Topics: Analytical Solutions, Analyzer, Water Chemistry, Waltron
Optimizing Combustion and Monitoring Tube Temperature for Safe and Efficient Processes
Topics: Analytical Solutions, AMETEK Process Instruments, Analyzer
Topics: Analytical Solutions, Analyzer, KECO, New Mexico
Controlling the world’s water resources is arguably one of the most critical issues. Water demand from industry and domestic users is set to rise throughout the industrialized world, while water supplies are finite. Legislation constantly demands improvements in potable water quality and reduces the contaminants that may be discharged. Wastewater treatment is one way to get water back. Wastewater is subjected to various treatments in wastewater treatment plants, each intended to remove contaminants and other hazardous substances from the water. The objective is to create water from sewage that is safe to discharge into waterways and won’t endanger the environment or human health. Over the last few years, Yokogawa has been applying minimized maintenance measurement systems to an industry that, more than ever, is concerned with condition-based maintenance and the integrity of the measured variable.
Topics: Yokogawa, Analytical Solutions, Wastewater, Analyzer
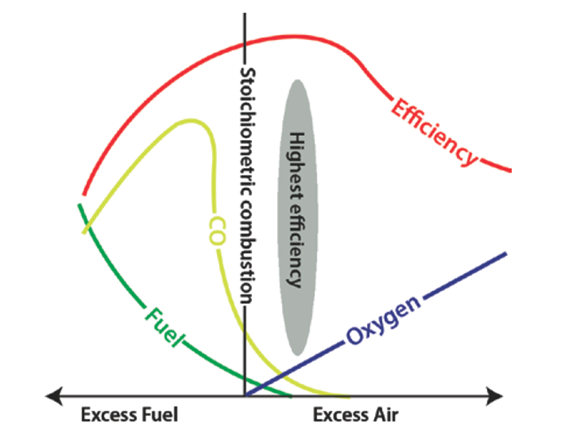
In any combustion process, it is necessary to monitor the inlet flow rates of fuel and air to the burner. While these flow rates provide baseline parameters to set a flame, they do not provide feedback to reveal or alert any potential concerns with the combustion reaction, such as incomplete combustion from imperfect mixing in the burner or safety risks such as fuel leaks or loss of flame. Flue gas analysis offers one approach to monitor the process and provide feedback. It is especially important to consider when firing both hydrocarbon-based and high hydrogen fuels.
Topics: Analytical Solutions, AMETEK Process Instruments, Analyzer
If you work in the natural gas industry or are an industrial user of natural gas, water dewpoint is not only a quality measurement but also a big concern. Water condensate presents serious challenges and issues for your process. It is highly corrosive and will form hydrates, which are ice-like solid molecules that can block the flow of gas in pipelines. In cold weather, it will also freeze reducing the pipe pressure.
Topics: Moisture Measurement Solutions, Analytical Solutions, AMETEK Process Instruments, Analyzer
Optimizing Boiler Efficiency | Increase Your Bottom Line
Optimizing boiler efficiency will increase your bottom line. Did you know that every 10% excess oxygen results in 1% in wasted fuel? With that in mind, Yokogawa Zirconia Oxygen analyzers have a 50-year proven success record for providing:
Topics: Yokogawa, Analytical Solutions, Analyzer